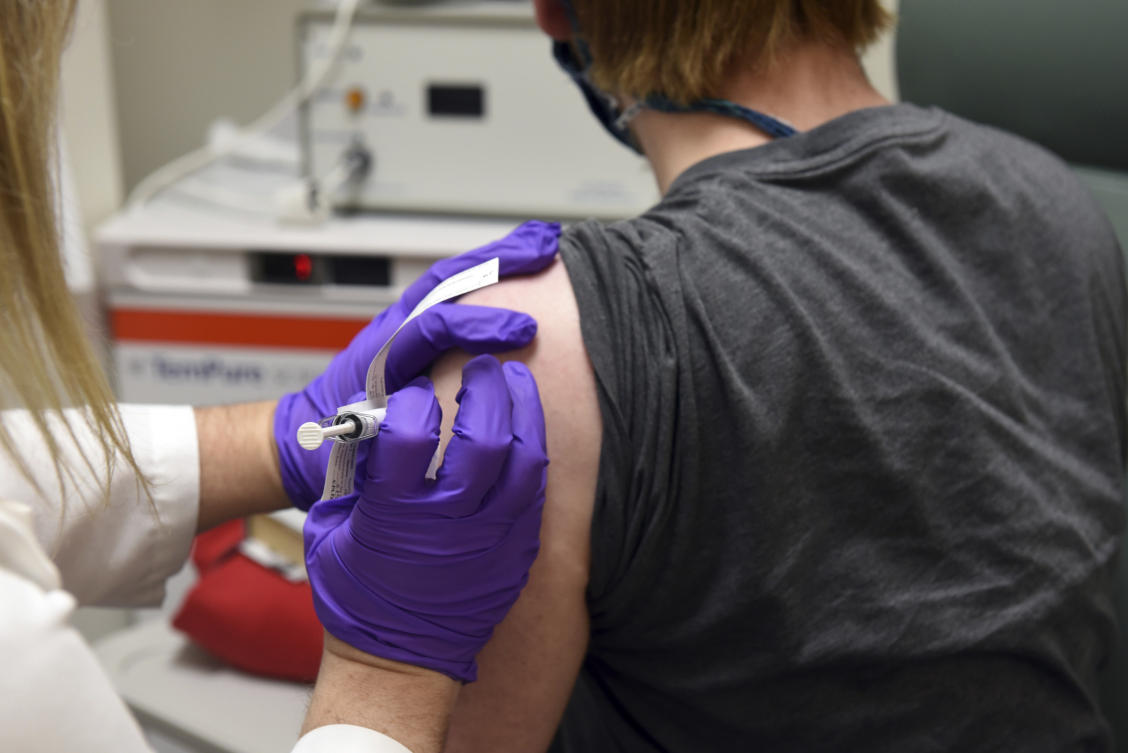
WASHINGTON: US pharmaceutical giant Pfizer expects to file for emergency use authorization for its Covid-19 vaccine in late November, around two weeks after the November 3 US presidential election,
Full Story
Follow Arabian Post
Select Arabian Post as your preferred source on Google and MSN News for trusted business news and Arab politics and updates.
Notice an issue?
Arabian Post strives to deliver the most accurate and reliable information to its readers. If you believe you have identified an error or inconsistency in this article, please don't hesitate to contact our editorial team at editor[at]thearabianpost[dot]com. We are committed to promptly addressing any concerns and ensuring the highest level of journalistic integrity.







